 
What did you observe? Give explanations for your observations. Add two drops of concentrated nitric acid and immediately put on the lid. Finally, put one piece of copper turning into the Petri dish.Add two drops of dilute sulfuric acid to each metal in the sulfuric acid column.Add two drops of dilute nitric acid to each metal in the nitric acid column.Add two drops of dilute hydrochloric acid to each metal in the hydrochloric acid column.When all the pieces of metal are in place: Finally, place a few tin granules in each box in the tin row.Place some iron filings in each box in the iron row.Place a few zinc granules in each box in the zinc row.Place one small piece of magnesium ribbon in each box in the magnesium row.Place a few copper turnings in each box in the copper row.Cover the table on your worksheet with a clear plastic sheet.Zinc powder, Zn(s), is FLAMMABLE and hazardous to the aquatic environment – see CLEAPSS Hazcard HC107.Īs you do these experiments observe carefully and record your findings.Magnesium ribbon is FLAMMABLE – see CLEAPSS Hazcard HC059A.Sulfuric acid, H 2SO 4(aq), 1 mol dm –3 is CORROSIVE – see CLEAPSS Hazcard HC098a and CLEAPSS Recipe Book RB098.Concentrated nitric acid, HNO 3(aq), 5 mol dm –3 is OXIDISING, CORROSIVE and gives off TOXIC fumes – see CLEAPSS Hazcard HC067 and CLEAPSS Recipe Book RB061.Dilute nitric acid, HNO 3(aq), 1 mol dm –3 is CORROSIVE – see CLEAPSS Hazcard HC067 and CLEAPSS Recipe Book RB061.Hydrochloric acid, HCl(aq), 1 mol dm –3 is low hazard – see CLEAPSS Hazcard HC047a and CLEAPSS Recipe Book RB043.Do NOT increase quantities mentioned and work in a well-ventilated laboratory. Nitrogen oxides are formed, gases are CORROSIVE and very TOXIC (fatal if inhaled) – see CLEAPSS Hazcard HC068B.Wear eye protection throughout (splash-resistant goggles to BS EN166 3).Read our standard health and safety guidance.See the accompanying guidance on apparatus and techniques for microscale chemistry, which includes instructions for preparing a variety of solutions. Solutions should be contained in plastic pipettes. RSC Yusuf Hamied Inspirational Science Programme.Introductory maths for higher education.The physics of restoration and conservation.The electrochemical series as a series of metals arranged in order of their ability to be oxidised (reactions, other than displacement reactions, not required).Investigate the properties of different materials including solubilities, conductivity, melting points and boiling points. Unit C2: Further Chemical Reactions, Rates and Equilibrium, Calculations and Organic Chemistry.2.1.4 explain and describe the displacement reactions of metals with other metal ions in solution.2.1.1 recall the reactivity series of metals, including K, Na, Ca, Mg, Al, Zn, Fe and Cu.Unit 2: Further Chemical Reactions, Rates and Equilibrium, Calculations and Organic Chemistry.make reasoned judgements and draw evidence-based conclusions.PRACTICAL: Determination of relative reactivities of metals through displacement reactions.iron nail in copper(II) chloride solution) and competition reactions (e.g. (c) the relative reactivities of metals as demonstrated by displacement (e.g.Unit 2: CHEMICAL BONDING, APPLICATION OF CHEMICAL REACTIONS and ORGANIC CHEMISTRY.The method used to extract a metal from its ore depends on the position of the metal in the reactivity series.The electrochemical series represents a series of reduction reactions.C3.2.1 deduce an order of reactivity of metals based on experimental results including reactions with water, dilute acid and displacement reactions with other metals.C3.2 How are metals with different reactivities extracted?.C3 Chemicals of the natural environment. 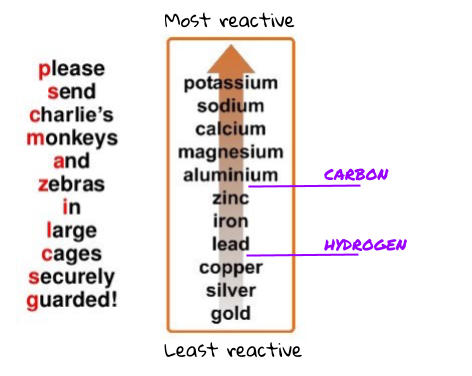
10 Investigate the variables that affect temperature changes in reacting solutions such as, eg acid plus metals, acid plus carbonates, neutralisations, displacement of metals.RP18 Investigate the variables that affect the temperature changes of a series of reactions in solutions, eg acid plus metals, acid plus carbonates, neutralisations, displacement of metals.A more reactive metal can displace a less reactive metal from a compound. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
